News
OrganVision: Cardiac tissue engineering – Visualizing
The human heart pumps ~70 ml blood per beat which is ~5 l per minute. Cardiomyocytes (heart muscle cells) are the working horse in this system. In the heart cardiomyocytes are organized in a complex 3-dimensional tissue. They are functionally and structurally coupled to variety of other cell types, e.g. endothelial cells, fibroblasts, vascular smooth muscle cells, but also immune cells such as macrophages. Even though they make up most of the heart`s volume only 20% to 30% of the heart`s cells are cardiomyocytes. Cardiac tissue engineering was established to study tissue organization and heart muscle function in a 3D environment in a dish.
Initially rodent heart muscle cells have been used but over the last ten years progress in stem cell biology enabled us to use human cardiomyocytes. Human pluripotent (embryonic or induced pluripotent) stem cells can be efficiently differentiated to cardiomyocytes. Pluripotent stem cell-derived cardiomyocytes (Figure 1) can then be used to engineer three-dimensional cardiac tissue constructs (engineered heart tissue; EHT). EHTs are three-dimensional tissue constructs in which stem cell-derived cardiomyocytes are embedded in a hydrogel matrix. Stem cell derived cardiomyocytes are immature but mature during culture in this three-dimensional environment (Figure 2). After ~5-10 days in culture EHTs coherently start to beat (Movie 1) and can be used to study heart muscle function under normal and pathological conditions in a dish.
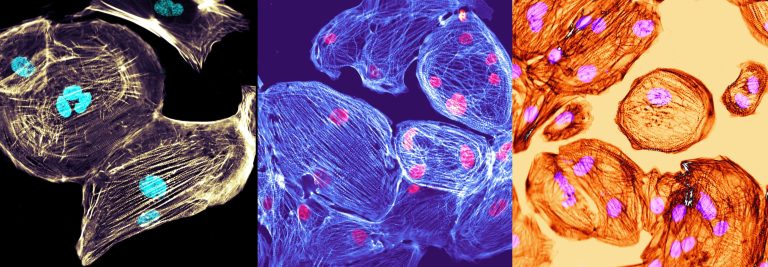
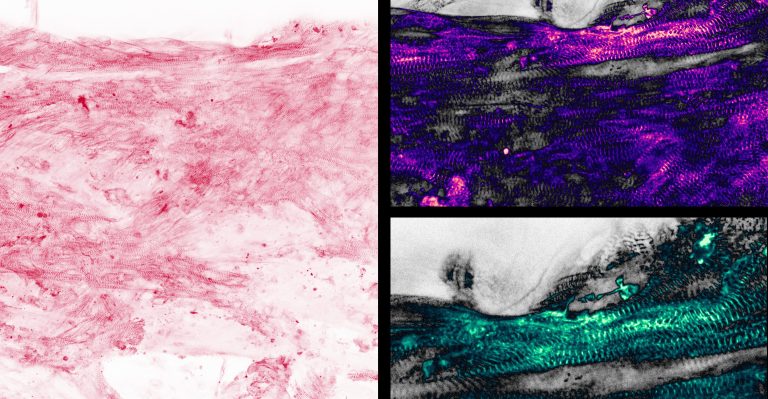
Whereas functional analysis has been successfully implemented morphological analysis was only possible after the EHTs have been harvested. OrganVision aims to develop techniques to visualize tissue development and function in real-time. We will include stem-cell derived non-myocytes (that make up most of the heart`s cells and are a lot more than just passive bystanders) and study cell–cell interactions as well as heart muscle function under normal and pathological conditions. The project will be conducted by PhD-student Nancy Shehata and Dr. Florian Weinberger.
Recent Posts
- Decoding multiple scattering from microscopy data January 2, 2024
- It rained innovation awards at JenLab: Celebrating a Year of Innovation at JenLab! December 5, 2023
- OrganVision: WP6 August 30, 2023
- OrganVision: WP6 August 22, 2023
- OrganVision: WP8 July 26, 2023
